
SVN-001
About SVN-001
SVN-001 is a Phase 3 investigational, novel combination therapy consisting of an N-methyl-D-aspartate ("NMDA") receptor-modulating drug (ketamine) delivered intravenously ("IV") in combination with copywritten relapse prevention cognitive behavioural therapy ("CBT") for the treatment of severe AUD targeting the UK and EU markets.
A successful Phase 2a/b trial demonstrated a 50% reduction in heavy drinking days vs a placebo and 86% abstinence on average sustained for six months post-treatment, compared to just 2% pre-trial.
Treatment cycle
Three IV sessions along with structured CBT across 12 weeks; practical for outpatient delivery.
Week 1
Initial medical assessment hour long initial assessment (IMA)
Prep session with client & therapist 90 mins
Ketamine 1
Integration 1
Week 2
Prep session 2
Ket session 2
Integration session 2
Week 3
Prep session 3
Ket session 3
Integration session 3
Close out session - with Dr. Therapist & Patient
Clinical evidence
Phase 2 results demonstrated a 50% reduction in heavy drinking days vs a placebo.
Long-term impact: 86% abstinence on average sustained for six months post-treatment, compared to just 2% pre-trial. Patients also saw improvements in liver function, depression and quality of life, with no drug-related SAEs.
Regulatory pathway
A full mixed marketing authorisation in the UK and EU is planned, which, if successful, will provide regulatory protection to include eight years of data exclusivity in the EU and ten years of concurrent market exclusivity in both regions.
Clinical development
SVN-001 is in phase 3, with an n=280 two-armed active placebo-controlled ‘More Kare’ trial. It is co-funded by the UK National Institute for Health Research Efficacy and Mechanism Evaluation Programme (NIHR150193) and Solvonis. It is being run by the University of Exeter Clinical Trials Unit.
SVN-001
SVN-001 Phase III Estimated Trial Design
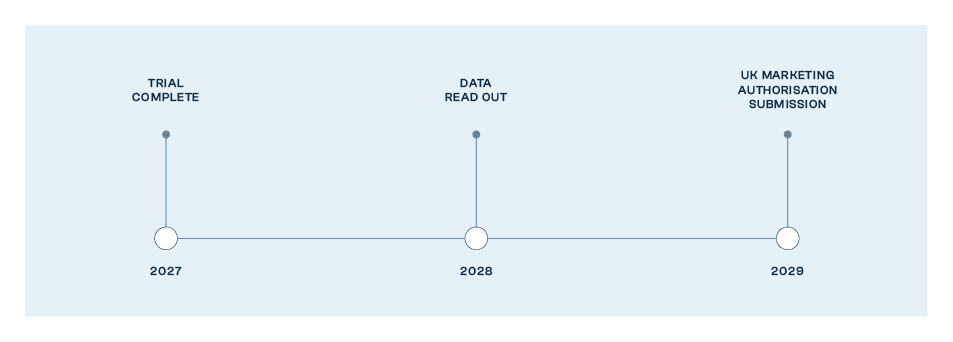
SVN-001 Ph3 trial design
n=280 two-armed placebo-controlled trial.
Trial co-funded by UK Dept of Health and Social Care.
Nine NHS Trust Sites.
Trial started Q3 2024.
MHRA innovative passport (ILAP) awarded.
SVN-001 Phase III Trial Design
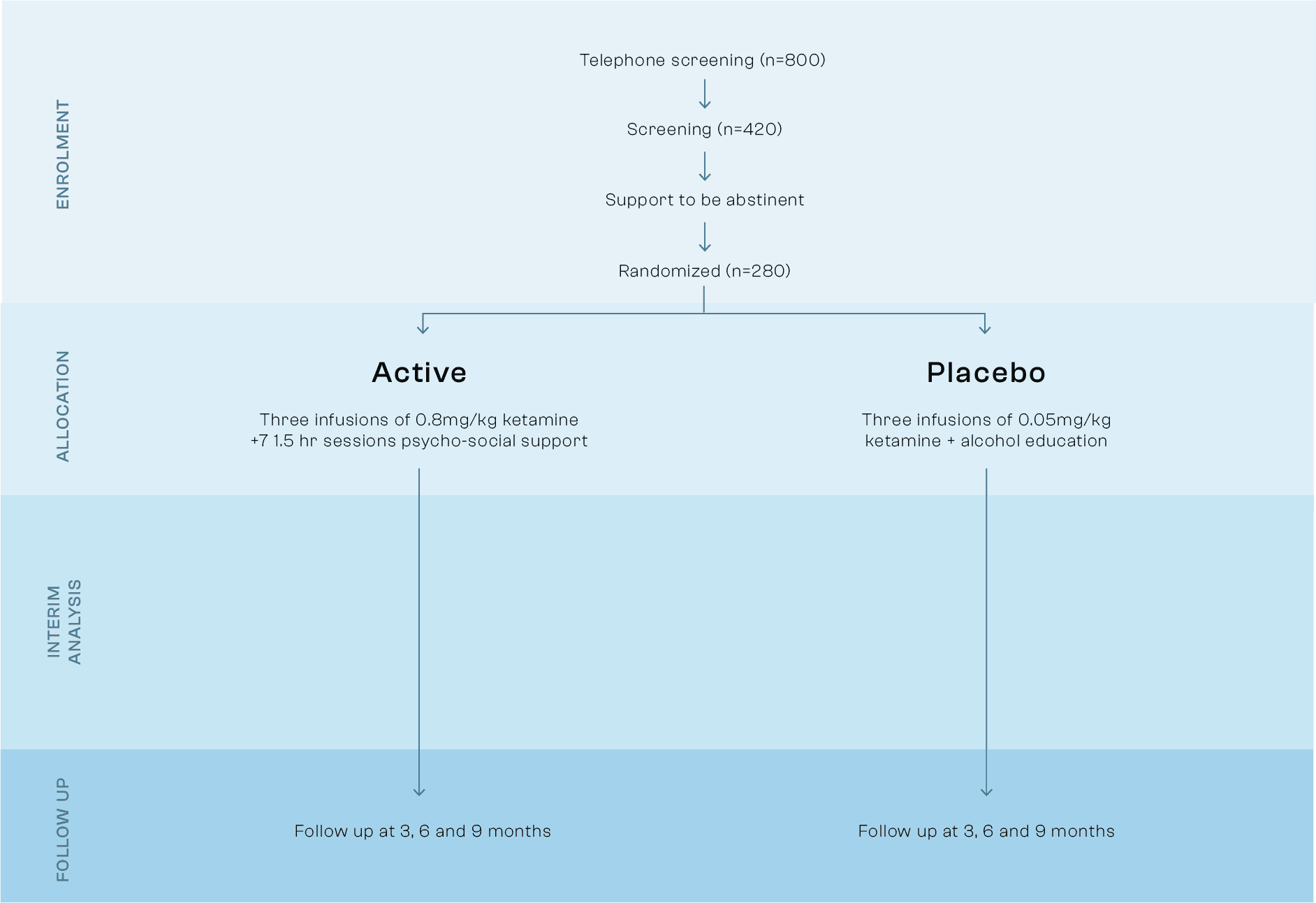
Ph2 results
Primary Outcome: Days Abstinent
The trial met its primary success criteria by achieving a prolonged and statistically significantly increase in rates of abstinence in the six months post treatment. With 86% abstinence observed in the KARE therapy arm compared to 70% in the placebo and alcohol education arm.

Primary Outcome: Days Abstinent
With 86% abstinence observed in the KARE therapy arm, this is more than double the 38% abstinence observed in a similar group of patients receiving industry standard treatment as usual in the US. The KARE therapy produced a statistically significant increase in percentage days abstinent in comparison to the placebo and education group.
* Treatment as usual specialist outpatient alcohol care in US taken from a trial (Oslin et al. 2013) was chosen as a descriptive comparator as KARE will likely be delivered in outpatient settings.
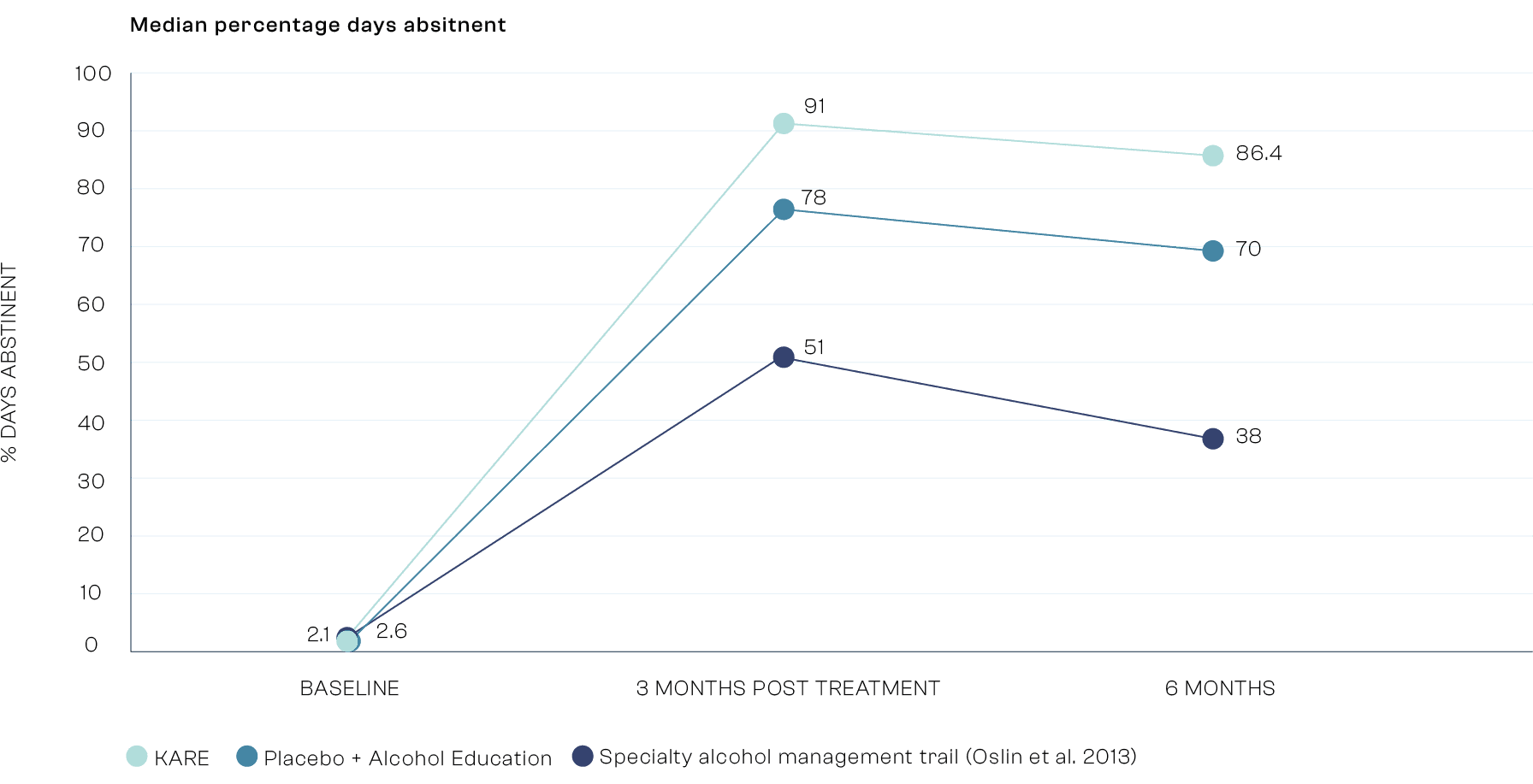
Co-Primary Outcome: Relapse
The KARE group were 2.1 more likely than the placebo education group and 2.5 times more likely than those in the industry standard treatment as usual study to have no Heavy Drinking Days in the 6 months after treatment. Heavy Drinking Days, defined as 65g of alcohol for men, 52g for women in a single day, is an FDA and EMA regulatory endpoint guideline.
![Bar chart comparing the probability of relapse versus no relapse (P[Relapse] / P[No Relapse]) across three groups. The y-axis is labelled ‘P(Relapse) / P(No Relapse)’, ranging from 0 to 4. The x-axis shows three groups: 1. KARE (light teal) 2. Placebo + Alcohol Education (medium teal) 3. Specialty Alcohol Management Trial (Oslin et al. 2013) (dark blue) Values: • KARE group: 1.2 with an error bar ranging from approximately 0.8 to 1.6 • Placebo + Alcohol Education: 3.16 with a visible error bar • Oslin et al. 2013 group: 3.0 with a visible error bar The chart indicates a lower relapse probability in the KARE group compared to the other two groups.](https://framerusercontent.com/images/rz2P5ND5BUNNyO8ZHEKX0qjhDhI.png?width=1792&height=917)
* Treatment as usual specialist outpatient alcohol care in US taken from a trial (Oslin et al. 2013) as KARE will likely be delivered in outpatient settings.
Exploratory Analysis: Number of Days Heavy Drinking
While not one of the trials primary endpoints, we saw a statistically significant reduction in 'Heavy Drinking Days' when comparing KARE to placebo and education group. When comparing KARE to the industry standard treatment as usual in the US, we can see 77% reduction in Heavy Drinking Days. Heavy Drinking Days is a primary endpoint used by EMA and FDA when assessing marketing authorization/ regulatory approval.
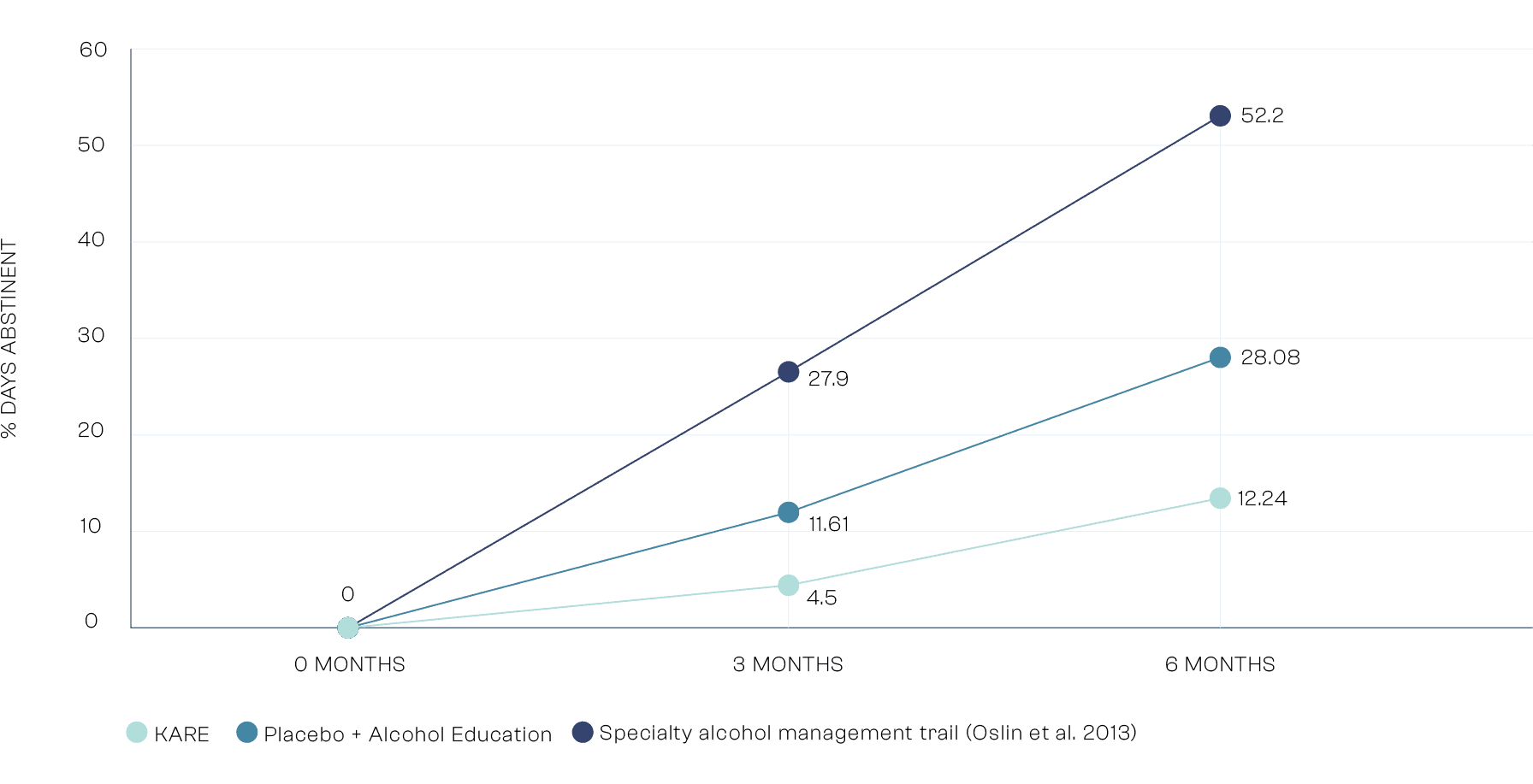
* Treatment as usual specialist outpatient alcohol care in US taken from a trial (Oslin et al. 2013) as KARE will likely be delivered in outpatient settings.
Secondary Outcomes: Depression
We excluded people who were taking anti-depressant medication at the time of the trial. This resulted in a minimally depressed sample with lower depression than a typical group of patients with Alcohol Use Disorder. There was a statistically significantly reduction in depression in the ketamine groups vs placebo groups at 3 months. This shows that ketamine is an effective antidepressant in patients with Alcohol Use Disorder.
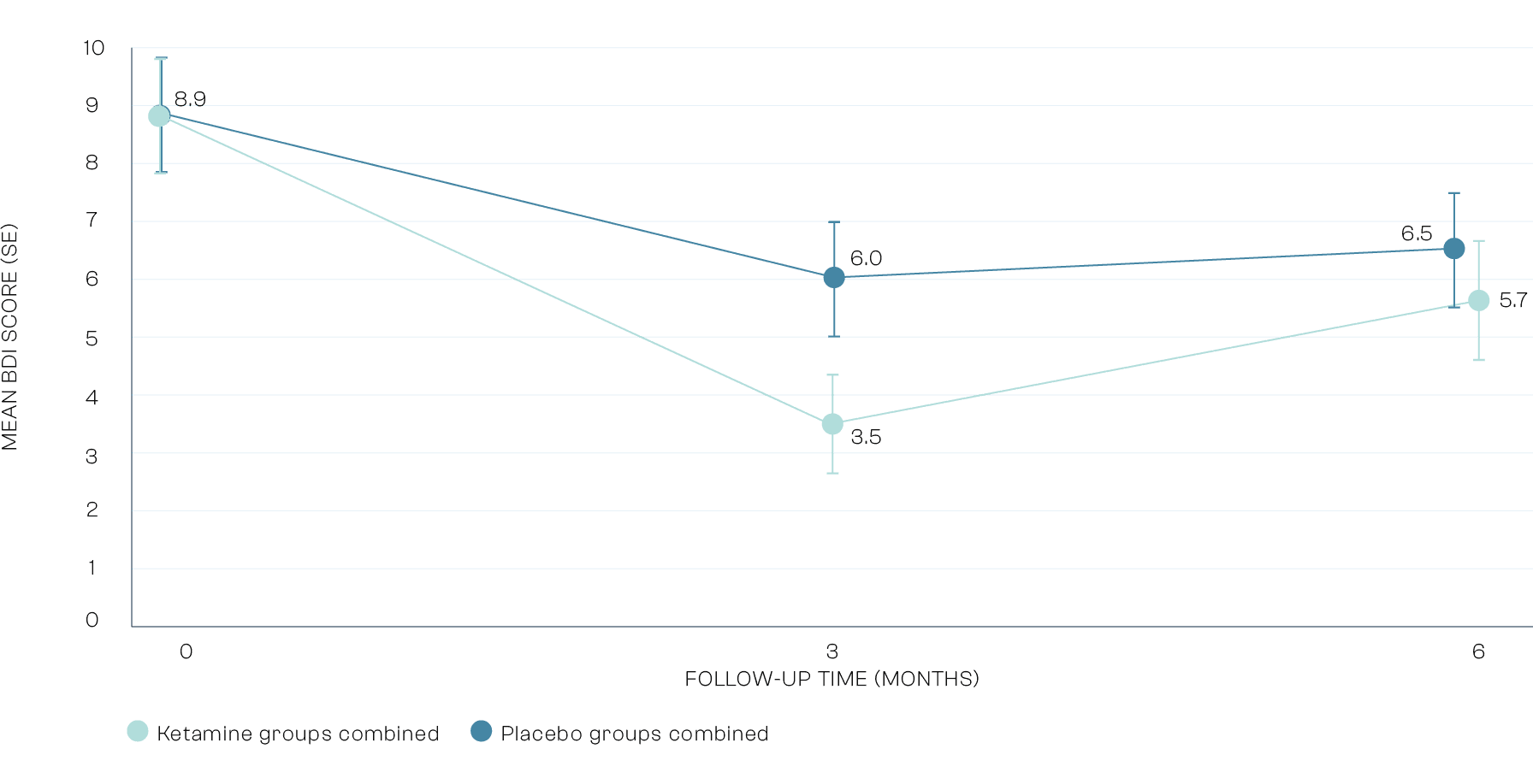
* Treatment as usual specialist outpatient alcohol care in US taken from a trial (Oslin et al. 2013) as KARE will likely be delivered in outpatient settings.
Secondary Outcomes: Liver Function
There was a statistically significantly improvement in liver function at 6 months post trial. This was measured across 4 different validated biomarkers of liver function. Our data indicates that ketamine is not only safe for the treatment of Alcohol Use Disorder, but is also associated with an improvement in liver function. Liver disease is the now second most common cause of preventable death in the UK.
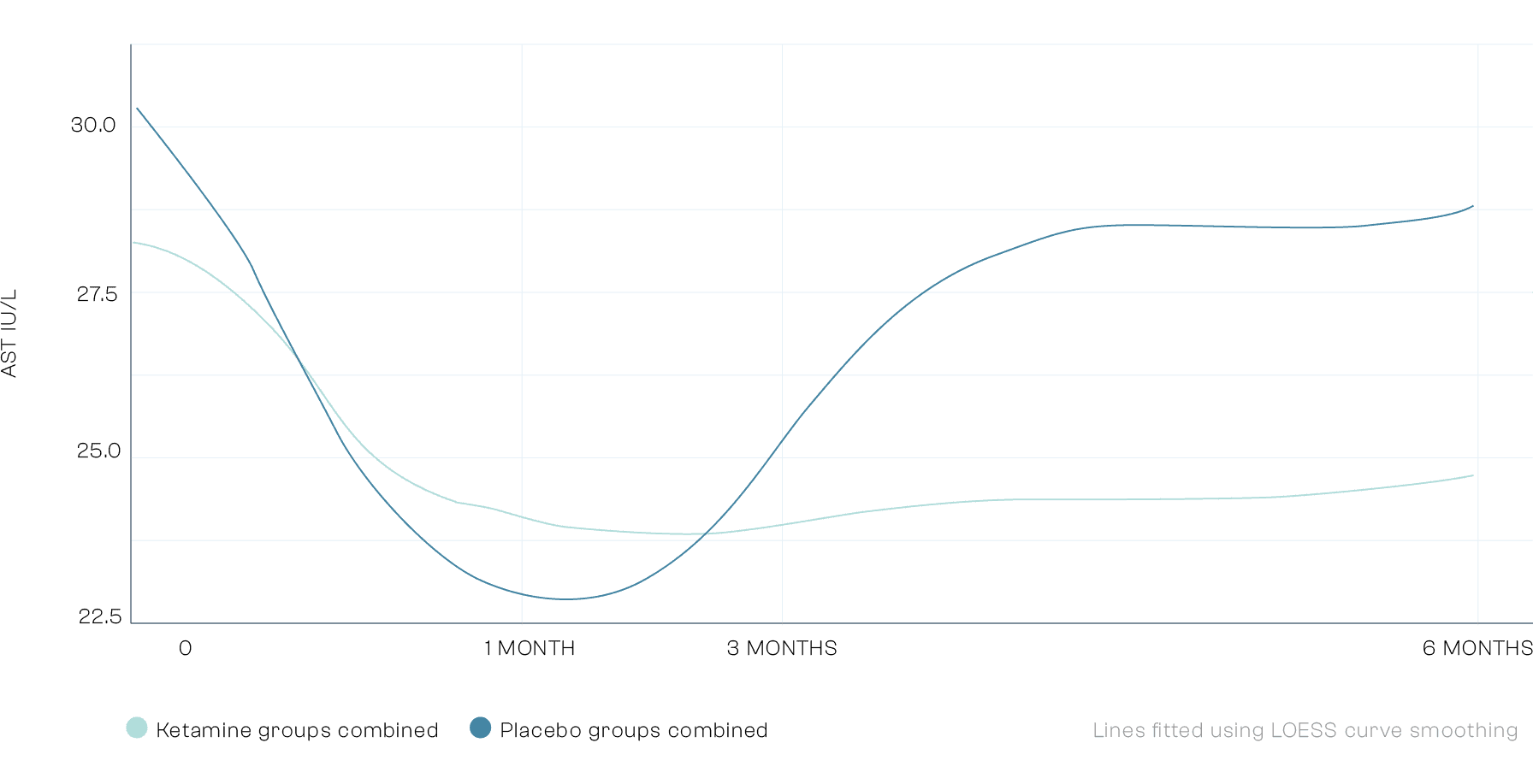
Secondary Outcomes: Anhedonia
Anhedonia which is the inability to experience pleasure, is common across a number of mental health problems. Ketamine helped to increase the ability to experience pleasure. The impact of ketamine on anhedonia shows how the treatment goes beyond reducing alcohol consumption.
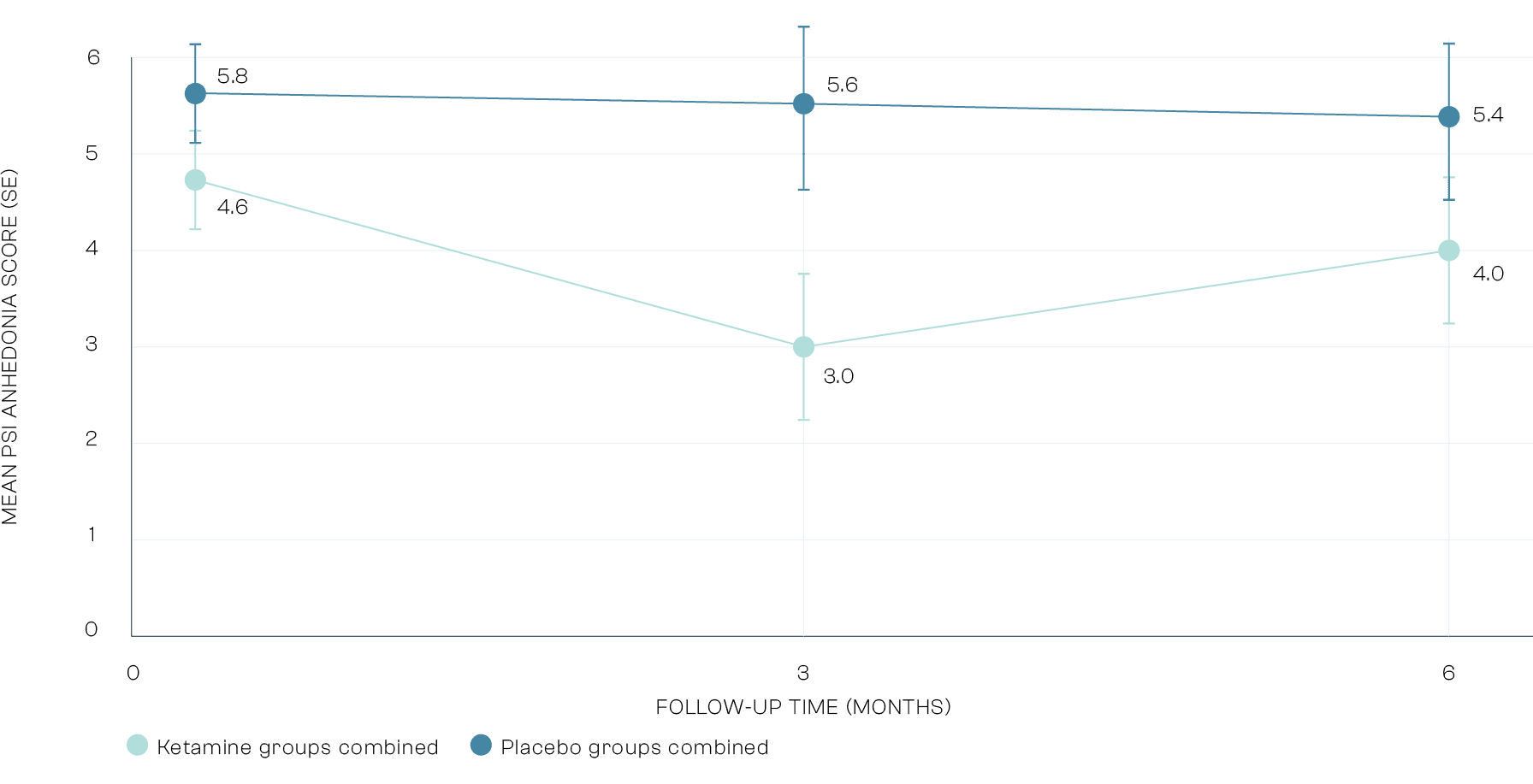
Primary Outcome: Days Abstinent
The trial met its primary success criteria by achieving a prolonged and statistically significantly increase in rates of abstinence in the six months post treatment. With 86% abstinence observed in the KARE therapy arm compared to 70% in the placebo and alcohol education arm.

Primary Outcome: Days Abstinent
With 86% abstinence observed in the KARE therapy arm, this is more than double the 38% abstinence observed in a similar group of patients receiving industry standard treatment as usual in the US. The KARE therapy produced a statistically significant increase in percentage days abstinent in comparison to the placebo and education group.

* Treatment as usual specialist outpatient alcohol care in US taken from a trial (Oslin et al. 2013) was chosen as a descriptive comparator as KARE will likely be delivered in outpatient settings.
Co-Primary Outcome: Relapse
The KARE group were 2.1 more likely than the placebo education group and 2.5 times more likely than those in the industry standard treatment as usual study to have no Heavy Drinking Days in the 6 months after treatment. Heavy Drinking Days, defined as 65g of alcohol for men, 52g for women in a single day, is an FDA and EMA regulatory endpoint guideline.
![Bar chart comparing the probability of relapse versus no relapse (P[Relapse] / P[No Relapse]) across three groups. The y-axis is labelled ‘P(Relapse) / P(No Relapse)’, ranging from 0 to 4. The x-axis shows three groups: 1. KARE (light teal) 2. Placebo + Alcohol Education (medium teal) 3. Specialty Alcohol Management Trial (Oslin et al. 2013) (dark blue) Values: • KARE group: 1.2 with an error bar ranging from approximately 0.8 to 1.6 • Placebo + Alcohol Education: 3.16 with a visible error bar • Oslin et al. 2013 group: 3.0 with a visible error bar The chart indicates a lower relapse probability in the KARE group compared to the other two groups.](https://framerusercontent.com/images/rz2P5ND5BUNNyO8ZHEKX0qjhDhI.png?width=1792&height=917)
* Treatment as usual specialist outpatient alcohol care in US taken from a trial (Oslin et al. 2013) as KARE will likely be delivered in outpatient settings.
Exploratory Analysis: Number of Days Heavy Drinking
While not one of the trials primary endpoints, we saw a statistically significant reduction in 'Heavy Drinking Days' when comparing KARE to placebo and education group. When comparing KARE to the industry standard treatment as usual in the US, we can see 77% reduction in Heavy Drinking Days. Heavy Drinking Days is a primary endpoint used by EMA and FDA when assessing marketing authorization/ regulatory approval.

* Treatment as usual specialist outpatient alcohol care in US taken from a trial (Oslin et al. 2013) as KARE will likely be delivered in outpatient settings.
Secondary Outcomes: Depression
We excluded people who were taking anti-depressant medication at the time of the trial. This resulted in a minimally depressed sample with lower depression than a typical group of patients with Alcohol Use Disorder. There was a statistically significantly reduction in depression in the ketamine groups vs placebo groups at 3 months. This shows that ketamine is an effective antidepressant in patients with Alcohol Use Disorder.

* Treatment as usual specialist outpatient alcohol care in US taken from a trial (Oslin et al. 2013) as KARE will likely be delivered in outpatient settings.
Secondary Outcomes: Liver Function
There was a statistically significantly improvement in liver function at 6 months post trial. This was measured across 4 different validated biomarkers of liver function. Our data indicates that ketamine is not only safe for the treatment of Alcohol Use Disorder, but is also associated with an improvement in liver function. Liver disease is the now second most common cause of preventable death in the UK.

Secondary Outcomes: Anhedonia
Anhedonia which is the inability to experience pleasure, is common across a number of mental health problems. Ketamine helped to increase the ability to experience pleasure. The impact of ketamine on anhedonia shows how the treatment goes beyond reducing alcohol consumption.

Primary Outcome: Days Abstinent
The trial met its primary success criteria by achieving a prolonged and statistically significantly increase in rates of abstinence in the six months post treatment. With 86% abstinence observed in the KARE therapy arm compared to 70% in the placebo and alcohol education arm.

Primary Outcome: Days Abstinent
With 86% abstinence observed in the KARE therapy arm, this is more than double the 38% abstinence observed in a similar group of patients receiving industry standard treatment as usual in the US. The KARE therapy produced a statistically significant increase in percentage days abstinent in comparison to the placebo and education group.

* Treatment as usual specialist outpatient alcohol care in US taken from a trial (Oslin et al. 2013) was chosen as a descriptive comparator as KARE will likely be delivered in outpatient settings.
Co-Primary Outcome: Relapse
The KARE group were 2.1 more likely than the placebo education group and 2.5 times more likely than those in the industry standard treatment as usual study to have no Heavy Drinking Days in the 6 months after treatment. Heavy Drinking Days, defined as 65g of alcohol for men, 52g for women in a single day, is an FDA and EMA regulatory endpoint guideline.
![Bar chart comparing the probability of relapse versus no relapse (P[Relapse] / P[No Relapse]) across three groups. The y-axis is labelled ‘P(Relapse) / P(No Relapse)’, ranging from 0 to 4. The x-axis shows three groups: 1. KARE (light teal) 2. Placebo + Alcohol Education (medium teal) 3. Specialty Alcohol Management Trial (Oslin et al. 2013) (dark blue) Values: • KARE group: 1.2 with an error bar ranging from approximately 0.8 to 1.6 • Placebo + Alcohol Education: 3.16 with a visible error bar • Oslin et al. 2013 group: 3.0 with a visible error bar The chart indicates a lower relapse probability in the KARE group compared to the other two groups.](https://framerusercontent.com/images/rz2P5ND5BUNNyO8ZHEKX0qjhDhI.png?width=1792&height=917)
* Treatment as usual specialist outpatient alcohol care in US taken from a trial (Oslin et al. 2013) as KARE will likely be delivered in outpatient settings.
Exploratory Analysis: Number of Days Heavy Drinking
While not one of the trials primary endpoints, we saw a statistically significant reduction in 'Heavy Drinking Days' when comparing KARE to placebo and education group. When comparing KARE to the industry standard treatment as usual in the US, we can see 77% reduction in Heavy Drinking Days. Heavy Drinking Days is a primary endpoint used by EMA and FDA when assessing marketing authorization/ regulatory approval.

* Treatment as usual specialist outpatient alcohol care in US taken from a trial (Oslin et al. 2013) as KARE will likely be delivered in outpatient settings.
Secondary Outcomes: Depression
We excluded people who were taking anti-depressant medication at the time of the trial. This resulted in a minimally depressed sample with lower depression than a typical group of patients with Alcohol Use Disorder. There was a statistically significantly reduction in depression in the ketamine groups vs placebo groups at 3 months. This shows that ketamine is an effective antidepressant in patients with Alcohol Use Disorder.

* Treatment as usual specialist outpatient alcohol care in US taken from a trial (Oslin et al. 2013) as KARE will likely be delivered in outpatient settings.
Secondary Outcomes: Liver Function
There was a statistically significantly improvement in liver function at 6 months post trial. This was measured across 4 different validated biomarkers of liver function. Our data indicates that ketamine is not only safe for the treatment of Alcohol Use Disorder, but is also associated with an improvement in liver function. Liver disease is the now second most common cause of preventable death in the UK.

Secondary Outcomes: Anhedonia
Anhedonia which is the inability to experience pleasure, is common across a number of mental health problems. Ketamine helped to increase the ability to experience pleasure. The impact of ketamine on anhedonia shows how the treatment goes beyond reducing alcohol consumption.

Primary Outcome: Days Abstinent
The trial met its primary success criteria by achieving a prolonged and statistically significantly increase in rates of abstinence in the six months post treatment. With 86% abstinence observed in the KARE therapy arm compared to 70% in the placebo and alcohol education arm.

Primary Outcome: Days Abstinent
With 86% abstinence observed in the KARE therapy arm, this is more than double the 38% abstinence observed in a similar group of patients receiving industry standard treatment as usual in the US. The KARE therapy produced a statistically significant increase in percentage days abstinent in comparison to the placebo and education group.

* Treatment as usual specialist outpatient alcohol care in US taken from a trial (Oslin et al. 2013) was chosen as a descriptive comparator as KARE will likely be delivered in outpatient settings.
Co-Primary Outcome: Relapse
The KARE group were 2.1 more likely than the placebo education group and 2.5 times more likely than those in the industry standard treatment as usual study to have no Heavy Drinking Days in the 6 months after treatment. Heavy Drinking Days, defined as 65g of alcohol for men, 52g for women in a single day, is an FDA and EMA regulatory endpoint guideline.
![Bar chart comparing the probability of relapse versus no relapse (P[Relapse] / P[No Relapse]) across three groups. The y-axis is labelled ‘P(Relapse) / P(No Relapse)’, ranging from 0 to 4. The x-axis shows three groups: 1. KARE (light teal) 2. Placebo + Alcohol Education (medium teal) 3. Specialty Alcohol Management Trial (Oslin et al. 2013) (dark blue) Values: • KARE group: 1.2 with an error bar ranging from approximately 0.8 to 1.6 • Placebo + Alcohol Education: 3.16 with a visible error bar • Oslin et al. 2013 group: 3.0 with a visible error bar The chart indicates a lower relapse probability in the KARE group compared to the other two groups.](https://framerusercontent.com/images/rz2P5ND5BUNNyO8ZHEKX0qjhDhI.png?width=1792&height=917)
* Treatment as usual specialist outpatient alcohol care in US taken from a trial (Oslin et al. 2013) as KARE will likely be delivered in outpatient settings.
Exploratory Analysis: Number of Days Heavy Drinking
While not one of the trials primary endpoints, we saw a statistically significant reduction in 'Heavy Drinking Days' when comparing KARE to placebo and education group. When comparing KARE to the industry standard treatment as usual in the US, we can see 77% reduction in Heavy Drinking Days. Heavy Drinking Days is a primary endpoint used by EMA and FDA when assessing marketing authorization/ regulatory approval.

* Treatment as usual specialist outpatient alcohol care in US taken from a trial (Oslin et al. 2013) as KARE will likely be delivered in outpatient settings.
Secondary Outcomes: Depression
We excluded people who were taking anti-depressant medication at the time of the trial. This resulted in a minimally depressed sample with lower depression than a typical group of patients with Alcohol Use Disorder. There was a statistically significantly reduction in depression in the ketamine groups vs placebo groups at 3 months. This shows that ketamine is an effective antidepressant in patients with Alcohol Use Disorder.

* Treatment as usual specialist outpatient alcohol care in US taken from a trial (Oslin et al. 2013) as KARE will likely be delivered in outpatient settings.
Secondary Outcomes: Liver Function
There was a statistically significantly improvement in liver function at 6 months post trial. This was measured across 4 different validated biomarkers of liver function. Our data indicates that ketamine is not only safe for the treatment of Alcohol Use Disorder, but is also associated with an improvement in liver function. Liver disease is the now second most common cause of preventable death in the UK.

Secondary Outcomes: Anhedonia
Anhedonia which is the inability to experience pleasure, is common across a number of mental health problems. Ketamine helped to increase the ability to experience pleasure. The impact of ketamine on anhedonia shows how the treatment goes beyond reducing alcohol consumption.

Primary Outcome: Days Abstinent
The trial met its primary success criteria by achieving a prolonged and statistically significantly increase in rates of abstinence in the six months post treatment. With 86% abstinence observed in the KARE therapy arm compared to 70% in the placebo and alcohol education arm.

Primary Outcome: Days Abstinent
With 86% abstinence observed in the KARE therapy arm, this is more than double the 38% abstinence observed in a similar group of patients receiving industry standard treatment as usual in the US. The KARE therapy produced a statistically significant increase in percentage days abstinent in comparison to the placebo and education group.

* Treatment as usual specialist outpatient alcohol care in US taken from a trial (Oslin et al. 2013) was chosen as a descriptive comparator as KARE will likely be delivered in outpatient settings.
Co-Primary Outcome: Relapse
The KARE group were 2.1 more likely than the placebo education group and 2.5 times more likely than those in the industry standard treatment as usual study to have no Heavy Drinking Days in the 6 months after treatment. Heavy Drinking Days, defined as 65g of alcohol for men, 52g for women in a single day, is an FDA and EMA regulatory endpoint guideline.
![Bar chart comparing the probability of relapse versus no relapse (P[Relapse] / P[No Relapse]) across three groups. The y-axis is labelled ‘P(Relapse) / P(No Relapse)’, ranging from 0 to 4. The x-axis shows three groups: 1. KARE (light teal) 2. Placebo + Alcohol Education (medium teal) 3. Specialty Alcohol Management Trial (Oslin et al. 2013) (dark blue) Values: • KARE group: 1.2 with an error bar ranging from approximately 0.8 to 1.6 • Placebo + Alcohol Education: 3.16 with a visible error bar • Oslin et al. 2013 group: 3.0 with a visible error bar The chart indicates a lower relapse probability in the KARE group compared to the other two groups.](https://framerusercontent.com/images/rz2P5ND5BUNNyO8ZHEKX0qjhDhI.png?width=1792&height=917)
* Treatment as usual specialist outpatient alcohol care in US taken from a trial (Oslin et al. 2013) as KARE will likely be delivered in outpatient settings.
Exploratory Analysis: Number of Days Heavy Drinking
While not one of the trials primary endpoints, we saw a statistically significant reduction in 'Heavy Drinking Days' when comparing KARE to placebo and education group. When comparing KARE to the industry standard treatment as usual in the US, we can see 77% reduction in Heavy Drinking Days. Heavy Drinking Days is a primary endpoint used by EMA and FDA when assessing marketing authorization/ regulatory approval.

* Treatment as usual specialist outpatient alcohol care in US taken from a trial (Oslin et al. 2013) as KARE will likely be delivered in outpatient settings.
Secondary Outcomes: Depression
We excluded people who were taking anti-depressant medication at the time of the trial. This resulted in a minimally depressed sample with lower depression than a typical group of patients with Alcohol Use Disorder. There was a statistically significantly reduction in depression in the ketamine groups vs placebo groups at 3 months. This shows that ketamine is an effective antidepressant in patients with Alcohol Use Disorder.

* Treatment as usual specialist outpatient alcohol care in US taken from a trial (Oslin et al. 2013) as KARE will likely be delivered in outpatient settings.
Secondary Outcomes: Liver Function
There was a statistically significantly improvement in liver function at 6 months post trial. This was measured across 4 different validated biomarkers of liver function. Our data indicates that ketamine is not only safe for the treatment of Alcohol Use Disorder, but is also associated with an improvement in liver function. Liver disease is the now second most common cause of preventable death in the UK.

Secondary Outcomes: Anhedonia
Anhedonia which is the inability to experience pleasure, is common across a number of mental health problems. Ketamine helped to increase the ability to experience pleasure. The impact of ketamine on anhedonia shows how the treatment goes beyond reducing alcohol consumption.

Primary Outcome: Days Abstinent
The trial met its primary success criteria by achieving a prolonged and statistically significantly increase in rates of abstinence in the six months post treatment. With 86% abstinence observed in the KARE therapy arm compared to 70% in the placebo and alcohol education arm.

Primary Outcome: Days Abstinent
With 86% abstinence observed in the KARE therapy arm, this is more than double the 38% abstinence observed in a similar group of patients receiving industry standard treatment as usual in the US. The KARE therapy produced a statistically significant increase in percentage days abstinent in comparison to the placebo and education group.

* Treatment as usual specialist outpatient alcohol care in US taken from a trial (Oslin et al. 2013) was chosen as a descriptive comparator as KARE will likely be delivered in outpatient settings.
Co-Primary Outcome: Relapse
The KARE group were 2.1 more likely than the placebo education group and 2.5 times more likely than those in the industry standard treatment as usual study to have no Heavy Drinking Days in the 6 months after treatment. Heavy Drinking Days, defined as 65g of alcohol for men, 52g for women in a single day, is an FDA and EMA regulatory endpoint guideline.
![Bar chart comparing the probability of relapse versus no relapse (P[Relapse] / P[No Relapse]) across three groups. The y-axis is labelled ‘P(Relapse) / P(No Relapse)’, ranging from 0 to 4. The x-axis shows three groups: 1. KARE (light teal) 2. Placebo + Alcohol Education (medium teal) 3. Specialty Alcohol Management Trial (Oslin et al. 2013) (dark blue) Values: • KARE group: 1.2 with an error bar ranging from approximately 0.8 to 1.6 • Placebo + Alcohol Education: 3.16 with a visible error bar • Oslin et al. 2013 group: 3.0 with a visible error bar The chart indicates a lower relapse probability in the KARE group compared to the other two groups.](https://framerusercontent.com/images/rz2P5ND5BUNNyO8ZHEKX0qjhDhI.png?width=1792&height=917)
* Treatment as usual specialist outpatient alcohol care in US taken from a trial (Oslin et al. 2013) as KARE will likely be delivered in outpatient settings.
Exploratory Analysis: Number of Days Heavy Drinking
While not one of the trials primary endpoints, we saw a statistically significant reduction in 'Heavy Drinking Days' when comparing KARE to placebo and education group. When comparing KARE to the industry standard treatment as usual in the US, we can see 77% reduction in Heavy Drinking Days. Heavy Drinking Days is a primary endpoint used by EMA and FDA when assessing marketing authorization/ regulatory approval.

* Treatment as usual specialist outpatient alcohol care in US taken from a trial (Oslin et al. 2013) as KARE will likely be delivered in outpatient settings.
Secondary Outcomes: Depression
We excluded people who were taking anti-depressant medication at the time of the trial. This resulted in a minimally depressed sample with lower depression than a typical group of patients with Alcohol Use Disorder. There was a statistically significantly reduction in depression in the ketamine groups vs placebo groups at 3 months. This shows that ketamine is an effective antidepressant in patients with Alcohol Use Disorder.

* Treatment as usual specialist outpatient alcohol care in US taken from a trial (Oslin et al. 2013) as KARE will likely be delivered in outpatient settings.
Secondary Outcomes: Liver Function
There was a statistically significantly improvement in liver function at 6 months post trial. This was measured across 4 different validated biomarkers of liver function. Our data indicates that ketamine is not only safe for the treatment of Alcohol Use Disorder, but is also associated with an improvement in liver function. Liver disease is the now second most common cause of preventable death in the UK.

Secondary Outcomes: Anhedonia
Anhedonia which is the inability to experience pleasure, is common across a number of mental health problems. Ketamine helped to increase the ability to experience pleasure. The impact of ketamine on anhedonia shows how the treatment goes beyond reducing alcohol consumption.

Primary Outcome: Days Abstinent
The trial met its primary success criteria by achieving a prolonged and statistically significantly increase in rates of abstinence in the six months post treatment. With 86% abstinence observed in the KARE therapy arm compared to 70% in the placebo and alcohol education arm.

Primary Outcome: Days Abstinent
With 86% abstinence observed in the KARE therapy arm, this is more than double the 38% abstinence observed in a similar group of patients receiving industry standard treatment as usual in the US. The KARE therapy produced a statistically significant increase in percentage days abstinent in comparison to the placebo and education group.

* Treatment as usual specialist outpatient alcohol care in US taken from a trial (Oslin et al. 2013) was chosen as a descriptive comparator as KARE will likely be delivered in outpatient settings.
Co-Primary Outcome: Relapse
The KARE group were 2.1 more likely than the placebo education group and 2.5 times more likely than those in the industry standard treatment as usual study to have no Heavy Drinking Days in the 6 months after treatment. Heavy Drinking Days, defined as 65g of alcohol for men, 52g for women in a single day, is an FDA and EMA regulatory endpoint guideline.
![Bar chart comparing the probability of relapse versus no relapse (P[Relapse] / P[No Relapse]) across three groups. The y-axis is labelled ‘P(Relapse) / P(No Relapse)’, ranging from 0 to 4. The x-axis shows three groups: 1. KARE (light teal) 2. Placebo + Alcohol Education (medium teal) 3. Specialty Alcohol Management Trial (Oslin et al. 2013) (dark blue) Values: • KARE group: 1.2 with an error bar ranging from approximately 0.8 to 1.6 • Placebo + Alcohol Education: 3.16 with a visible error bar • Oslin et al. 2013 group: 3.0 with a visible error bar The chart indicates a lower relapse probability in the KARE group compared to the other two groups.](https://framerusercontent.com/images/rz2P5ND5BUNNyO8ZHEKX0qjhDhI.png?width=1792&height=917)
* Treatment as usual specialist outpatient alcohol care in US taken from a trial (Oslin et al. 2013) as KARE will likely be delivered in outpatient settings.
Exploratory Analysis: Number of Days Heavy Drinking
While not one of the trials primary endpoints, we saw a statistically significant reduction in 'Heavy Drinking Days' when comparing KARE to placebo and education group. When comparing KARE to the industry standard treatment as usual in the US, we can see 77% reduction in Heavy Drinking Days. Heavy Drinking Days is a primary endpoint used by EMA and FDA when assessing marketing authorization/ regulatory approval.

* Treatment as usual specialist outpatient alcohol care in US taken from a trial (Oslin et al. 2013) as KARE will likely be delivered in outpatient settings.
Secondary Outcomes: Depression
We excluded people who were taking anti-depressant medication at the time of the trial. This resulted in a minimally depressed sample with lower depression than a typical group of patients with Alcohol Use Disorder. There was a statistically significantly reduction in depression in the ketamine groups vs placebo groups at 3 months. This shows that ketamine is an effective antidepressant in patients with Alcohol Use Disorder.

* Treatment as usual specialist outpatient alcohol care in US taken from a trial (Oslin et al. 2013) as KARE will likely be delivered in outpatient settings.
Secondary Outcomes: Liver Function
There was a statistically significantly improvement in liver function at 6 months post trial. This was measured across 4 different validated biomarkers of liver function. Our data indicates that ketamine is not only safe for the treatment of Alcohol Use Disorder, but is also associated with an improvement in liver function. Liver disease is the now second most common cause of preventable death in the UK.

Secondary Outcomes: Anhedonia
Anhedonia which is the inability to experience pleasure, is common across a number of mental health problems. Ketamine helped to increase the ability to experience pleasure. The impact of ketamine on anhedonia shows how the treatment goes beyond reducing alcohol consumption.

Primary Outcome: Days Abstinent
The trial met its primary success criteria by achieving a prolonged and statistically significantly increase in rates of abstinence in the six months post treatment. With 86% abstinence observed in the KARE therapy arm compared to 70% in the placebo and alcohol education arm.

Primary Outcome: Days Abstinent
With 86% abstinence observed in the KARE therapy arm, this is more than double the 38% abstinence observed in a similar group of patients receiving industry standard treatment as usual in the US. The KARE therapy produced a statistically significant increase in percentage days abstinent in comparison to the placebo and education group.

* Treatment as usual specialist outpatient alcohol care in US taken from a trial (Oslin et al. 2013) was chosen as a descriptive comparator as KARE will likely be delivered in outpatient settings.
Co-Primary Outcome: Relapse
The KARE group were 2.1 more likely than the placebo education group and 2.5 times more likely than those in the industry standard treatment as usual study to have no Heavy Drinking Days in the 6 months after treatment. Heavy Drinking Days, defined as 65g of alcohol for men, 52g for women in a single day, is an FDA and EMA regulatory endpoint guideline.
![Bar chart comparing the probability of relapse versus no relapse (P[Relapse] / P[No Relapse]) across three groups. The y-axis is labelled ‘P(Relapse) / P(No Relapse)’, ranging from 0 to 4. The x-axis shows three groups: 1. KARE (light teal) 2. Placebo + Alcohol Education (medium teal) 3. Specialty Alcohol Management Trial (Oslin et al. 2013) (dark blue) Values: • KARE group: 1.2 with an error bar ranging from approximately 0.8 to 1.6 • Placebo + Alcohol Education: 3.16 with a visible error bar • Oslin et al. 2013 group: 3.0 with a visible error bar The chart indicates a lower relapse probability in the KARE group compared to the other two groups.](https://framerusercontent.com/images/rz2P5ND5BUNNyO8ZHEKX0qjhDhI.png?width=1792&height=917)
* Treatment as usual specialist outpatient alcohol care in US taken from a trial (Oslin et al. 2013) as KARE will likely be delivered in outpatient settings.
Exploratory Analysis: Number of Days Heavy Drinking
While not one of the trials primary endpoints, we saw a statistically significant reduction in 'Heavy Drinking Days' when comparing KARE to placebo and education group. When comparing KARE to the industry standard treatment as usual in the US, we can see 77% reduction in Heavy Drinking Days. Heavy Drinking Days is a primary endpoint used by EMA and FDA when assessing marketing authorization/ regulatory approval.

* Treatment as usual specialist outpatient alcohol care in US taken from a trial (Oslin et al. 2013) as KARE will likely be delivered in outpatient settings.
Secondary Outcomes: Depression
We excluded people who were taking anti-depressant medication at the time of the trial. This resulted in a minimally depressed sample with lower depression than a typical group of patients with Alcohol Use Disorder. There was a statistically significantly reduction in depression in the ketamine groups vs placebo groups at 3 months. This shows that ketamine is an effective antidepressant in patients with Alcohol Use Disorder.

* Treatment as usual specialist outpatient alcohol care in US taken from a trial (Oslin et al. 2013) as KARE will likely be delivered in outpatient settings.
Secondary Outcomes: Liver Function
There was a statistically significantly improvement in liver function at 6 months post trial. This was measured across 4 different validated biomarkers of liver function. Our data indicates that ketamine is not only safe for the treatment of Alcohol Use Disorder, but is also associated with an improvement in liver function. Liver disease is the now second most common cause of preventable death in the UK.

Secondary Outcomes: Anhedonia
Anhedonia which is the inability to experience pleasure, is common across a number of mental health problems. Ketamine helped to increase the ability to experience pleasure. The impact of ketamine on anhedonia shows how the treatment goes beyond reducing alcohol consumption.
